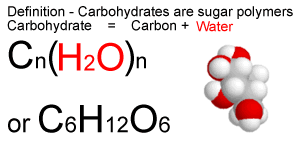
Starch is polymer os amylose and amylopectin. Starch: Part of the human diet, found in potato, rice, and cereal grain.Polysaccharide :Ĭarbohydrates consisting of large numbers of monosaccharides units linked through glycosidic bonds. Oligosaccharide is the polysaccharide of low molecular weight. Thus, d-glucose and d-mannose are epimeric at C-2 d-glucose and d-galactose are epimeric at C-4.ĭisaccharides are consisting of 2 monosaccharides joined covalently through an O-glycosidic bond that is formed when a -OH group of sugar reacts with anomeric carbon of the other. Sugars differing in configuration at a single asymmetric center are called epimers. These are a class of stereoisomers that are not mirror images of each other. Enantiomers:ĭ-Glyceraldehyde and l-glyceraldehyde are enantiomers or mirror images of each other. Glucose has 4 chiral carbons and 16 stereoisomers. ‘A molecule with n chiral centers can have 2n stereoisomers ’( Fisher projection) Two isomeric molecules having similar formula but differ in the three-dimensional orientation of their atoms in space. Two anomers are possible, but only the α anomer is shown. The open chain form of fructose gets converted into its cyclized form (to a five-membered ring) when the C-5 hydroxyl group attacks the C-2 ketone to form an intramolecular hemiketal. An anomeric carbon is a carbon derived from the carbonyl carbon of the open-chain form of the carbohydrate molecule. Two anomeric forms, designated α, and β can result. The open chain of glucose gets converted into its cyclized structure when the C-5 hydroxyl group attacks the oxygen atom of the C-1 aldehyde group to form an intramolecular hemiacetal. Five-member ring compounds are called furanosesand six members ring compounds are called Pyranoses. 
The formation of the cyclic ring is the result of a reaction between alcohol and aldehyde/ketone groups to form derivatives that are called Hemiacetals/Hemiketals. 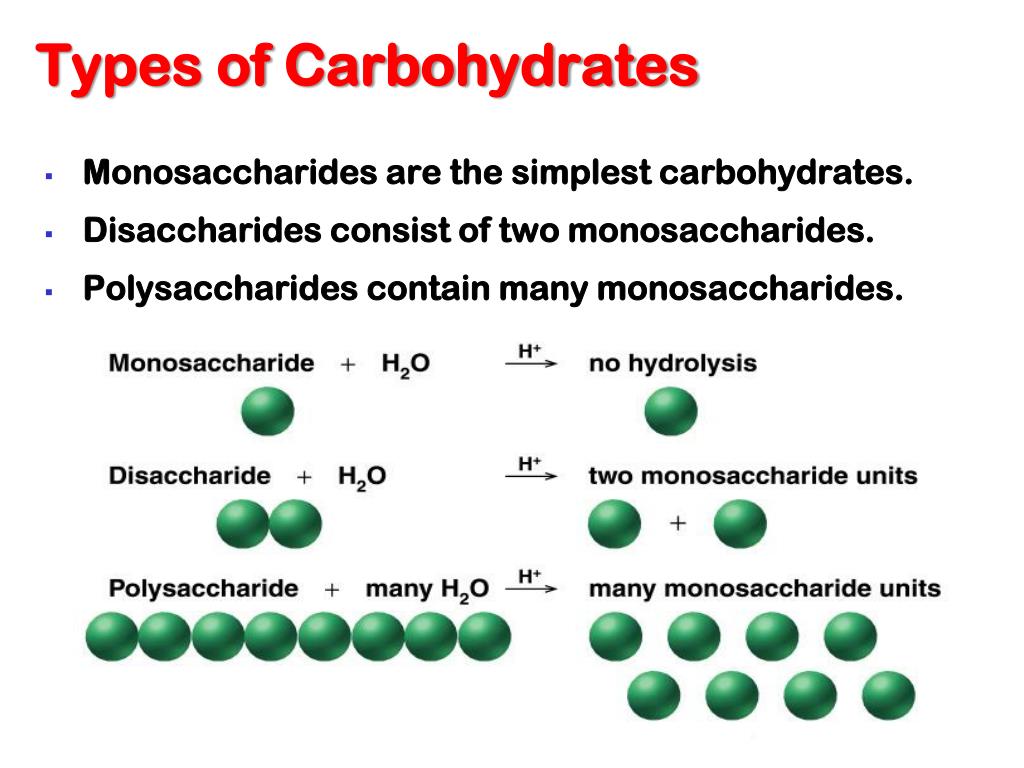
Monosaccharides are linked to alcohols and amines through glycosidic bonds.Most of the sugars are present in D form in nature except L-arabinose.It is the sweetest of all occurring sugars. Fructose is generally present in fruits, honey.Galactose is present in many plant gums and pectins and a component of disaccharide lactose.Excess glucose in our body is converted to fat and stored as adipose tissue. Excess glucose (Glycogen) is stored in the liver and muscle cells. Glucose is used in our body for energy production. Glucose is the most important monosaccharide.Monosaccharides having 3 carbon backbone are called Triose and four, five, six, and seven carbon backbone are called Tetroses, Pentose, Hexoses, Heptoses.If the carbonyl group of the sugar is attached at the end of the carbon chain the monosaccharide is aldoseand if the carbonyl group is at any other position, then the monosaccharide is a ketose.The symbols D and L designate the absolute configuration of the asymmetric carbon which is farthest from the aldehyde or keto group.Most of the monosaccharides have a sweet taste. Monosaccharides are crystalline solids, colorless, and freely soluble in water but insoluble in non-polar substances.Dihydroxyacetone acetone and L-glyceraldehyde is the smallest monosaccharides.Examples: Glucose, galactose, fructose, ribose.Most of the monosaccharides have the chemical formula (CH2O)n. Monosaccharides family starts with 3 carbon to 7 carbons backbones. 
The most abundant simple sugar found in nature is the six-carbon sugar D-glucose, also called dextrose. These are the building block for complex carbohydrates.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
